What is CGT
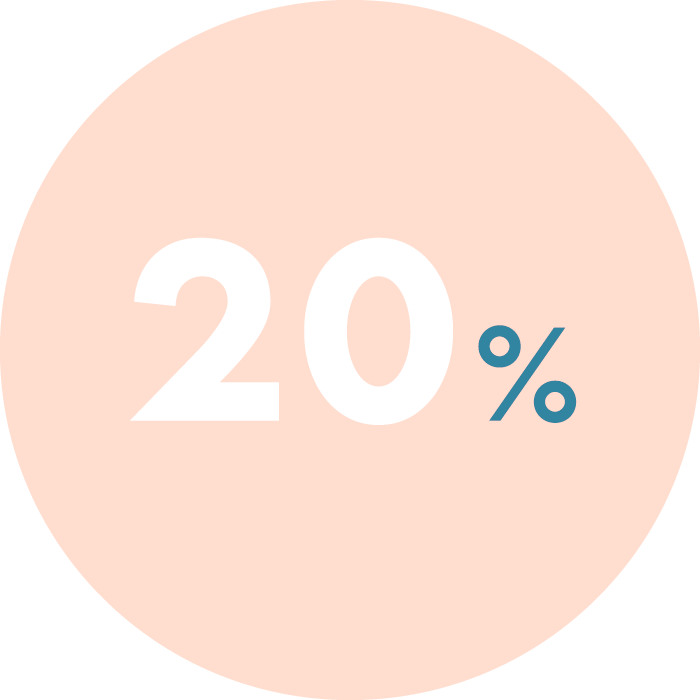
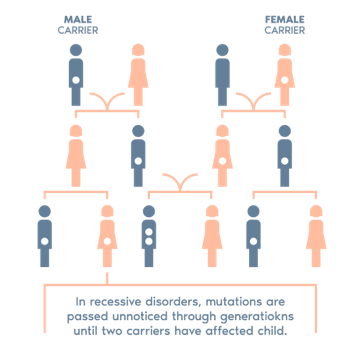
CGT is an advanced carrier genetic test, which determines whether a couple are carriers of genetic mutations that could be transmitted to their children.
CGT uses Next-Generation Sequencing (NGS) to analyse a wide panel of disorders including as Cystic Fibrosis, Spinal Muscular Atrophy, Fragile-X Syndrome.